Toxoplasma gondii: an opportunistic intracellular human parasite
Toxoplasma is an opportunistic pathogen with no cureToxoplasma gondii is a globally dominant, opportunistic human pathogen. Toxoplasma sets up a life-long chronic infection in its host that cannot be cured with currently available chemotherapies. Chronically infected humans that become immunocompromised due to HIV/AIDS are at risk to develop lethal toxoplasmosis.
|
Toxoplasma has a uniquely broad host rangeToxoplasma is related to other pathogens with a significant impact on human health, including the causative agents of malaria. Toxoplasma is unique in comparison to these other species in having a broader host range. For example, in addition to all mammals studied to date, Toxoplasma gondii can also infect and be transmitted by birds.
|
Ongoing research in the Boyle Lab
Below we describe two of the major projects going on in the laboratory. However we are addressing a number of other important questions in parasite biology, and are always open for graduate students and postdoctoral fellows to explore questions outside of these areas!!
Identification of secreted effectors in Toxoplasma gondii
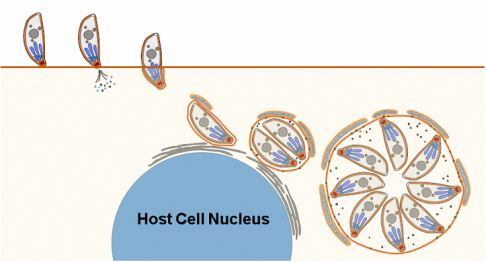
Toxoplasma must invade, and replicate within, its host cell. To do this Toxoplasma secretes multiple effector proteins during and after invasion.
We, and others in the Toxoplasma research community, have identified a handful of these effectors. For example, rhoptry proteins 5 and 18 dramatically alter virulence in the mouse model, and mitochondrial association factor 1 mediates the association of host mitochondrial with the parasite-containing vacuole.
In our lab we identify these effectors using a variety of bioinformatic and genetic approaches, determine their impact on parasite biology in vitro and in vivo using gene deletion/complementation experiments, and use biochemistry and genetics to elucidate their mode(s) of action.
We, and others in the Toxoplasma research community, have identified a handful of these effectors. For example, rhoptry proteins 5 and 18 dramatically alter virulence in the mouse model, and mitochondrial association factor 1 mediates the association of host mitochondrial with the parasite-containing vacuole.
In our lab we identify these effectors using a variety of bioinformatic and genetic approaches, determine their impact on parasite biology in vitro and in vivo using gene deletion/complementation experiments, and use biochemistry and genetics to elucidate their mode(s) of action.
Comparative and functional genomics of Toxoplasma and its near relatives
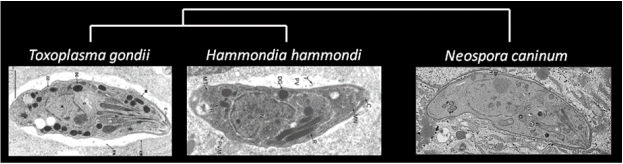
Toxoplasma is remarkable in having a vast host range; it is capable of infecting, causing disease in, and being transmitted by all warm-blooded animals. In contrast, its nearest extant relatives Hammondia hammondi and Neospora caninum do not share these life cycle features.
To identify the genes driving the unique life cycle features of T. gondii, we have taken a comparative approach. Using next-generation sequencing and comparative genomics we have determined that T. gondii and its nearest extant relative, Hammondia hammondi, share nearly all of their genes in near perfect synteny. Moreover, key virulence effectors (including ROP5 and ROP18) are conserved in both function and sequence between the two species.
We are currently using in vitro and in vivo techniques to:
The long-term goal of this work is to use these data to identify effectors and/or transcription factors that have led to the emergence of T. gondii as a formidable human pathogen.
To identify the genes driving the unique life cycle features of T. gondii, we have taken a comparative approach. Using next-generation sequencing and comparative genomics we have determined that T. gondii and its nearest extant relative, Hammondia hammondi, share nearly all of their genes in near perfect synteny. Moreover, key virulence effectors (including ROP5 and ROP18) are conserved in both function and sequence between the two species.
We are currently using in vitro and in vivo techniques to:
- Compare the host response to Toxoplasma, Hammondia hammondi and Neospora caninum in cell culture and during infections in mice.
- Identify parasite transcriptional networks that distinguish Toxoplasma and Hammondia hammondi.
- Adapt transgenic techniques that are effective in Toxoplasma for use in Hammondia hammondi.
The long-term goal of this work is to use these data to identify effectors and/or transcription factors that have led to the emergence of T. gondii as a formidable human pathogen.
